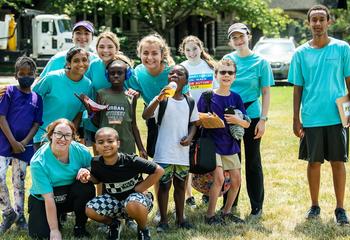
Senior Sarah Halupa, BS ’24, will graduate from Saint Joseph’s this spring and head to medical school at nearby Thomas Jefferson University, where she’ll join a program focused on providing better care for patients with autism.
Don't Miss Your Chance
Find your place. Break new ground. Here is where it all happens. Submit your enrollment deposit today.
Hundreds of degree options to fit your career path. Find yours now.
Saint Joseph’s University has a wide variety of bachelor's, master's, doctoral, associate or certificate programs. Here, you can choose a traditional academic path, combine majors across schools, opt for an accelerated or direct-entry pathway or enroll in a dual-degree program.
The University will award five honorary degrees at its 2024 Commencement ceremonies.

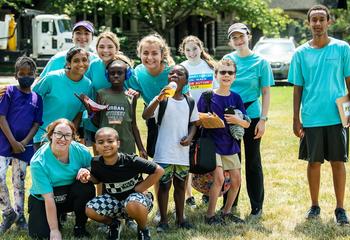
Senior Sarah Halupa, BS ’24, will graduate from Saint Joseph’s this spring and head to medical school at nearby Thomas Jefferson University, where she’ll join a program focused on providing better care for patients with autism.

Luca Toth, BS ’24, is the first student to graduate from ASPIRE – a college support program for students with autism spectrum disorder – with a full-time job lined up. In June, he’ll begin his new role as a business operations trainee with Philadelphia Insurance.

Ten reasons why Saint Joseph’s University should be at the top of your list when exploring your options for college.
Saint Joseph's five schools and colleges include an academic portfolio of more than 260 top-ranking programs in business, education, healthcare, arts and sciences and nursing.
As a graduate of the College of Arts and Sciences, you will be ready to engage the world around you and work with a sense of purpose. Through a combination of rigorous academics, experiential learning and guidance from dedicated faculty, you’ll be prepared with the skills, knowledge and confidence for success in a complex, fast-changing world.
The School of Education and Human Development challenges students to embrace the Jesuit concept of cura personalis — care for the whole person — in their professions in the way that they serve students, patients, clients and communities. We believe that the work of education and behavioral health professionals is essential to individual and community advancement. Careers in these sectors are more than jobs — they are driven by passion and purpose.
The Haub School of Business is committed to helping our students thrive in a world where technology-driven mindsets and models will change the nature of business. With our top-ranked business programs, we’re preparing graduates for an era where data analytics, artificial intelligence, digital supply chains, just-in-time learning and other new realities will present unprecedented challenges and opportunities. This unique approach doesn't go unnoticed, either. The Haub School of Business was recently named to Princeton Review's Best Business Schools and was ranked as a Tier One business school by CEO Magazine.
Whatever your path, the School of Health Professions offers a wide range of opportunities to help you reach your goals. Our programs feature a rigorous, established curriculum paired with the Jesuit, liberal arts core, as well as accelerated models for undergraduate students who want to pursue advanced careers in occupational therapy, physical therapy, pharmacy and physician assistant studies. Our School is also home to the Philadelphia College of Pharmacy, the first college of pharmacy in North America.
The School of Nursing and Allied Health at Saint Joseph's University offers a wide variety of bachelor's, master's, associate and certificate programs in specialized healthcare fields. You will benefit from a nursing school that has been a regional leader for over a century, with experienced faculty leading well-established coursework, simulation-based learning, and hands-on clinical experiences in Philadelphia, one of the nation’s healthcare hubs.
Saint Joseph's University nationally recognized centers and institutes are a hub of cross-disciplinary scholarship centered on community impact. From autism education to substance abuse disorder, bioethics to environmental stewardship, students and faculty bridge disciplines and enact real-world change through hands-on learning and research. Saint Joseph's students have the opportunity to combine classroom studies with exciting opportunities for hands-on learning led by experts in their fields.
There are countless opportunities to connect with the Hawk community, including joining one of our 100+ student organizations, an intramural sports team or one of our community service programs. Campus life is vibrant with the average Hawk joining three clubs during their time here.
